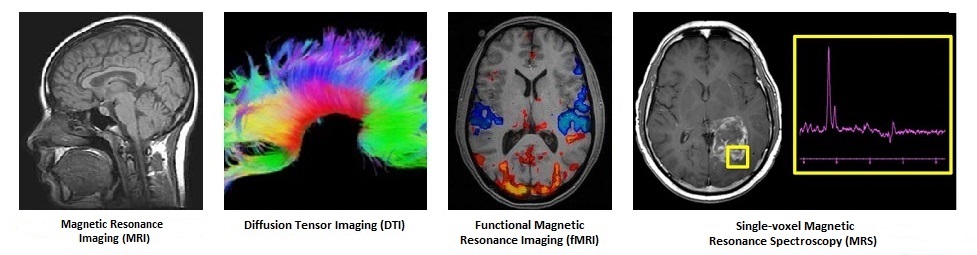
Imaging Biomarker for Addiction Treatment Outcome
*fMRI image courtesy of Courtesy Steve Smith, FMRIB This NIH IRB-approved study, which is being conducted by the Neuroimaging Research Branch (NRB) at NIDA in collaboration with the FSPHP, is recruiting 500 PHP (or PHP equivalent) participants. The goal of the research is to optimize the efficacy of currently available therapeutics by identifying brain biomarkers that can help assess the current severity of an individual’s substance use disorder (SUD), follow treatment progress and predict treatment outcome. Those who may be eligible for the study are:
The collaboration between NIDA and FSPHP helps ensure that this valuable research project can be completed with consideration for both research and quality clinical care. Together, we have established guidelines to assist PHP and Treatment Center staff with determining the appropriate methods for advertising the study. This study is approved by both the NIH IRB and the FSPHP research committee. Study Investigators, Coordinators, and Collaborators
How PHP Participants Can Get Involved
How Physician Health Programs Can Get Involved
How Treatment Centers Working with PHP Participants Can Get Involved
More Information about the Study
Contact Betty Jo Salmeron, M.D. directly at [email protected] (preferred) or 443-740-2651 to learn more about this unique, exciting, and vitally-important research study! |